Dielectric fluid properties
Test spectrum of the EPSILON+ in accordance with DIN 51111
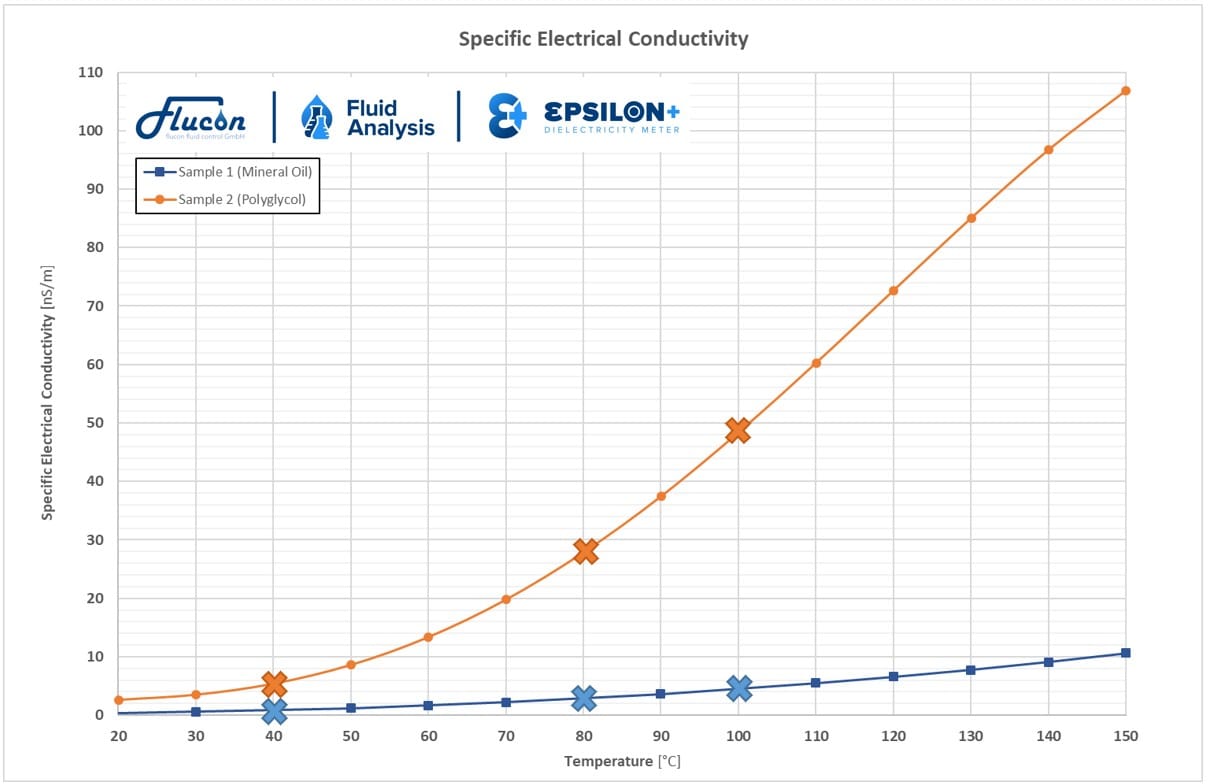
Specific electrical conductivity ϰ
The specific electrical conductivity, or conductivity, is a quantity which is often characterized by the symbol κ and indicates the ability of a substance to conduct electrical current. Its SI unit is S/m (Siemens per meter).
Our EPSILON+ Dielectricity Meter delivers measured values in in pS/m, nS/m and μS/m.
The suitability of a fluid to conduct electricity is determined by the number and mobility of the charge carriers within the liquid.
Specific electrical resistivity ρ
The specific electrical resistivity is the reciprocal of the specific electrical conductivity. It is often used for highly insulating liquids, such as transformer oils or immersion cooling fluids.
ρ = 1/ϰ .
The SI unit is Ω *m .
Dielectric dissipation factor tan δ (DDF)
The dielectric dissipation factor (DDF) or dielectric loss factor tan δ is a measure of the amount of energy that a substance in the alternating field absorbs and converts into heat loss. High dissipation factors are not suitable as insulating material in high frequency applications.
The loss factor is an indication of unwanted heat generation and thus provides information regarding the increased risk of thermal breakdown in transformer oils.
As can be seen in the following exemplary graphs, the electrical fluid properties are highly temperature-dependent:
Relative Permittivity / "Dielectric Constant" ε r
The relative permittivity, also known as "dielectric constant", was derived from the Latin term permittere = to let through. It is a measure of a liquid's permeability to electric fields.
The electric field constant ε 0, which equals the permittivity of the vacuum, serves as a factor for determination of the relative permittivity of a substance:
ε = ε 0 * ε r .
Here, ε r represents the substance-dependent relative permittivity. This is a dimensionless unit which is also temperature-dependent, as measurements with the EPSILON+ will show:

The all-new DIN 51111 test standard:
 The DIN 51 111 standard "Electrical properties of fresh and used oils from electric drives in vehicles – Measurement of the specific electrical conductivity, the relative permittivity (εr) and the dielectric dissipation factor (tan δ)" was published in February 2024.
The DIN 51 111 standard "Electrical properties of fresh and used oils from electric drives in vehicles – Measurement of the specific electrical conductivity, the relative permittivity (εr) and the dielectric dissipation factor (tan δ)" was published in February 2024.
It describes the electrical fluid characterization and thus the determination of the above-mentioned parameters as a function of the temperature by means of flucon's EPSILON+ tubular capacitor system.
For oils, the isotherms 50°C, 100°C and 150°C are recommended. Thus, the dry-block thermostat can be used with the EPSILON+.
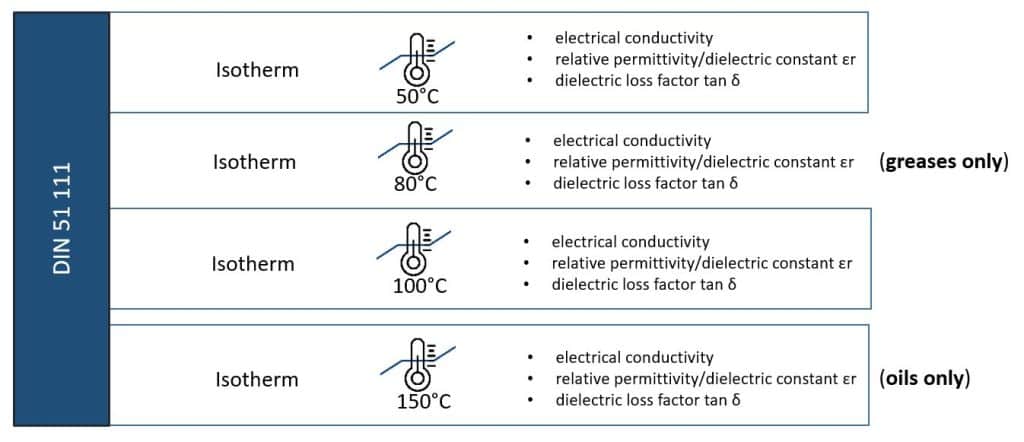
For the analysis of greases, on the other hand, the temperatures 50°C, 80°C, 100°C are recommended, as specified in M DIN 51 113.

